Article Text
Abstract
Objective To determine whether and how cognitive assessment data should be included in a report for patients with SLE and their providers.
Methods Leveraging experiences from prior studies, we created a cognitive report that included a hypothetical patient’s results on tests of multiple domains based on the NIH Toolbox Fluid Cognition Battery. In focus groups that comprised patients with SLE (two groups) and their providers (two groups), feedback was sought on the presentation of results as well as the potential value of the report in the clinical setting.
Results Feedback regarding the presentation of the report was generally positive. Both patients with SLE and their providers liked its simple graphics and use of a colour-gradated scale to indicate performance. However, both groups stressed the importance of using non-stigmatising language in describing results. Several potential purposes of the report, including distinguishing cognitive versus other issues, explaining cognitive challenges, improving patient–provider interactions, guiding decision-making, improving functioning or preventing impairment and tracking cognitive function over time, were noted by the participants. Potential barriers, such as inadequate clinical staffing or time and lack of potential treatments for identified issues, were also discussed.
Conclusion In this exploratory study, we found that both patients with SLE and their providers were receptive to the idea of a patient-friendly report of cognitive test results. This study provides important information to guide future pragmatic research to optimise the delivery of cognitive information to patients with SLE.
- systemic lupus erythematosus
- health services research
- outcome assessment
- health care
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
Key messages
What is already known about this subject?
Patients with SLE commonly report cognitive impairment, often described as ‘lupus fog’.
What does this study add?
This study explores patient and provider reactions to a patient-friendly report of cognitive functioning for SLE.
How might this impact on clinical practice or future developments?
The report could potentially be used by SLE patients and providers to identify and articulate cognitive problems, seek needed referrals and guide shared decision-making.
Introduction
Cognitive impairment in patients with SLE, often described as a ‘lupus fog’, has been reported in up to 50% of patients with SLE.1–6 This impairment may limit independence and reduce quality of life for patients with SLE, regardless of age. Despite its importance, cognitive functioning is not well described in most medical charts7 and often not discussed in the clinical setting. Specific tools to improve the recognition of cognitive impairment and facilitate its discussion are currently lacking in SLE.
To address this gap, we leveraged our experiences in (1) creating a physical functioning report from a previous study in patients receiving dialysis8 and (2) completing detailed cognitive assessments as part of a pilot study of functioning in patients with SLE (Approaches to Positive, Patient-centered Experiences of Aging in Lupus; APPEAL)9 to create a prototype cognitive functioning report for patients with SLE and obtain feedback on the value and presentation of the report. Our objective was to determine whether and how cognitive assessment data should be included in a functioning report for patients with SLE and their providers.
Methods
Study design and patient involvement
Four 90 min focus groups (two SLE patient (n=18) groups and two SLE provider groups (n=9)) were held in 2019. Patient participants were recruited from the APPEAL pilot,9 representing a subsample of the population-based Georgians Organized Against Lupus cohort10; provider participants (physicians, nurses, medical assistants) were recruited from the local provider population based on their experience with patients with SLE. While the initial prototype report was created by the study team, without patient involvement, these focus groups were designed to identify patient and provider priorities for the report of cognitive functioning information in future pragmatic research and ultimately clinical care.
Data collection and analysis
Development of the cognitive functioning report
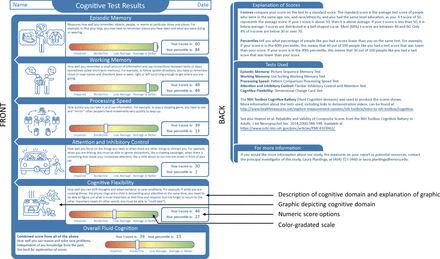
Based on a physical functioning report piloted in a population of patients receiving dialysis,8 our goal was to create a patient-friendly report for patients with SLE (figure 1). The domains of cognitive functioning included episodic memory, working memory, processing speed, inhibitory control and attention, and cognitive flexibility and were based on the NIH Toolbox11 assessments that were performed in the APPEAL pilot study.9 Everyday examples of tasks involving these cognitive domains were listed, discussed and selected by the entire team over the course of several meetings; the chosen examples were illustrated and provided along with a written description (figure 1). To gather feedback on preferred presentation of performance/scores, the hypothetical patient’s performance was displayed as both a colour-gradated scale with categories of ‘impaired’, ‘borderline’, ‘low average’ and ‘average or better’ and as numeric scores, including age, sex, race, ethnicity and education-adjusted t-scores (mean=50, SD=10) with associated percentiles. Further explanations were provided on the back of the report (figure 1).
Cognitive functioning report for a hypothetical patient with SLE, as presented to focus groups.
Focus groups
The focus groups included two phases: (1) an open discussion of cognitive problems and general interest in discussing cognition in clinic (Vandenberg et al, submitted) and (2) a discussion of the potential value of a cognitive report in a clinical setting as well as the specific presentation of domains and scores on the cognitive report. Focus group sessions were digitally recorded and transcribed. Transcripts were analysed (by AV) thematically12 with open coding,13 followed by focused coding using NVivo V.12 Plus (QSR International, Burlington, Massachusetts) data management and visualisation software.
Results
Focus group participants
The patient participants had a mean age of 51 and were predominantly female (83%) and Black (83%), and the mean duration of SLE was 20 years. The provider participants had a mean age of 40; 89% were female and 33% were Black. The provider participants reported a mean of 6 years of experience treating SLE.
Potential value of the cognitive report
Table 1 describes several stated potential purposes of having information about SLE patient cognition, such as that provided in the cognitive report. General comments included ‘I think it’s going to be really helpful’ (provider) and ‘I would love to do this with my doctor’ (patient). Specific to the report, both patients and providers voiced its potential to improve patient–provider interactions; patient participants envisioned the report as a communications tool: ‘It actually gives them (providers) now a visual of what it is that you may have been trying to express.’ While patient participants focused on the way that the report would initiate the conversation around cognition, provider participants focused more on how it would inform their communication with patients. In terms of guiding decision-making, both patients and provider participants mentioned follow-up referrals, and providers discussed the possibility of adjusting medications as needed.
Patient and provider feedback on potential value of shared information about cognition for patients with SLE
Patient and provider participants also had unique perspectives on the potential value and challenges of using the proposed report. First, patients less often mentioned the possibility of using the report to distinguish cognitive problems from other issues. Providers also stated that, even with cognitive test results, it might be impossible to distinguish the effects of age-related or dementia-related cognitive decline from the effects of SLE (including musculoskeletal problems that hinder testing, distracting pain, fatigue or depression). Second, several patient—but no provider—participants mentioned the possibility of using the report to identify cognitive problems for prevention of further deterioration or improvement. A related anticipated problem was the potential lack of a solution to the cognitive problems identified in the report (eg, ‘I would feel comfortable telling them (the results), as long as I had something for them to do to improve it’ (provider)). Finally, providers voiced significant concern about not having the time and resources to add cognitive assessment into clinic visits: ‘I’m not sure of how easy it would be in a clinic where I’m just the sole provider…if it was more of a multidisciplinary clinic, where you had nurses and social workers involved in the patient care, I think it would really work.’
Presentation of the cognitive report
The participants liked the overall appearance of the report, although a few patient participants suggested that the multiple results made it appear ‘complex’ at first glance. Feedback on graphic representations of cognitive domains was generally positive, with most participants understanding the tasks presented as relevant without the need for explanation. Table 2 presents feedback for the various presentations of the cognitive test results. Participants liked the simplicity of the colour-gradated scale, although some suggested changing the word ‘impaired’ to something less judgemental such as ‘fair’. In addition, most patient and some provider participants viewed the colour-gradated scale as a more neutral way to present results than numeric scores. Across groups, many suggested that the colour-gradated scale alone should be the primary presentation of results, potentially with numeric scores on the back of the report for providers and patients who wish to track these results. For numeric scores, most participants had preferences for percentiles over t-scores, if numeric scores were presented (table 1). While both represent comparisons to the general population, with adjustment for age, sex, race/ethnicity and education, some patients expressed preferences to be compared with a lupus population or not to be compared with others at all (eg, ‘The description says, “Like you.” I don't know what is like me…’).
Patient and provider feedback on performance reporting in a shared format cognitive report for patients with SLE
Discussion
In this exploratory study, we found that both patients with SLE and their providers were receptive to the idea of a patient-friendly report of cognitive test results. The report was seen by patients and providers as a tool to identify cognitive issues (or reassure patients of normal/stable functioning) and to facilitate better patient–provider interactions.
Importantly, this study, while limited by its qualitative nature and small sample size, identified several considerations for use of the report in the clinical or pragmatic research setting. First, expectations of patients with SLE regarding the potential for improvement in cognition should be managed. Few interventions to improve cognitive functioning have been studied in SLE to date; however, neither patients with SLE nor their providers mentioned the possibility of providing support (such as occupational therapy14) to help manage day-to-day tasks in the setting of cognitive impairment; education in this area of both patients and providers might be needed. Second, presentation of results is important; some patients with SLE may just want reassurance that functioning remains ‘average or better’, but other patients, and providers, may wish to track numeric results to identify longitudinal patterns. Third, the context of the clinical setting would have to be considered. Having providers on staff who feel comfortable discussing cognitive test results and have adequate time for the discussion is critical. Additionally, rheumatologists and primary care providers may wish to be aware of these issues to help guide their clinical decision-making, but they should also have the ability to refer a patient to a neurologist or other specialist specifically for further cognitive testing and/or to provide needed cognitive support. Partnership with geriatric providers could mitigate these issues by facilitating provider education and providing a cognitive support referral network.
In conclusion, these results provide important information to guide future pragmatic research to optimise the delivery of cognitive information to patients with SLE.
Acknowledgments
We thank Amelia Lambeth for creating the graphics used in our cognitive report.
Footnotes
Contributors LP, SSL, CBB and CD designed the study. LP, AV, FG, BJ, JJ and CBB were involved in the design and execution of the report prototype. LP, AV and CDT were involved in the acquisition of data. AV analysed the data. LP, AV, FG, CDT, BJ, JJ, SSL, CBB and CD reviewed the manuscript for intellectual content and approved the final manuscript.
Funding This work was supported by the US Army Medical Research Acquisition Activity (USAMRAA) through the Congressionally Directed Medical Research Programs (CDMRP) under Award No W81XWH1810619 (to LP). GOAL is supported by the Centers for Disease Control and Prevention (Grant No U01DP006488 to SSL and CD).
Disclaimer Opinions, interpretations, conclusions and recommendations are those of the authors and are not necessarily endorsed by the Department of Defense.
Competing interests None declared.
Patient and public involvement Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Patient consent for publication Not required.
Ethics approval The Emory Institutional Review Board and the Human Research Protection Office of the US Army Medical Research Acquisition Activity approved the study. All participants provided written informed consent.
Provenance and peer review Not commissioned; externally peer reviewed.