Abstract
The recognition that a multitude of interferon (IFN)-inducible genes are coordinately expressed in peripheral blood cells of patients with systemic lupus erythematosus (SLE) has contributed to considerable interest in the IFN pathway as a therapeutic target in lupus. Together with data that have accumulated over the past four decades implicat-ing IFN-á in SLE, the gene expression data have resulted in emergence of this cytokine pathway as a focal point for understanding mechanisms of autoimmunity and inflamma-tion in systemic autoimmune diseases. Assays that measure IFN-inducible gene expression in patient cells and tissues and plasma assays that quantify IFN-á protein are providing tools for identification of patients with active disease and who may be responsive to inhibition of the innate immune system component of the altered immune response in SLE. In addition, investigations of the mecha-nisms of induction of IFN pathway activation are suggesting clues to the triggers of autoimmunity in SLE.
Similar content being viewed by others
References and Recommended Reading
Pestka S, Krause CD, Walter MR: Interferons, interferon-like cytokines, and their receptors. Immunol Rev 2004, 202:38–32.
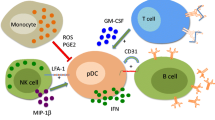
Ronnblom L, Alm GV: A pivotal role for the natural interferon a-producing cells (plasmacytoid dendritic cells) in the pathogenesis of lupus. J Exp Med 2001, 194:59–63. A comprehensive description of the complex scenario through which IFN-á might promote autoimmunity and disease in lupus.
Crow MK, Kirou KA: Interferon-alpha in systemic lupus erythematosus. Curr Opin Rheumatol 2004, 16:541–547. A review of the potential mechanisms that might account for IFN pathway activation in SLE.
Hooks JJ, Moutsopoulos HM, Geis SA, et al.: Immune interferon in the circulation of patients with autoimmune disease. N Engl J Med 1979, 301:5–8.
Hooks JJ, Jordan GW, Cupps T, et al.: Multiple interferons in the circulation of patients with systemic lupus erythematosus and vasculitis. Arthritis Rheum 1982, 25:396–400.
Preble OT, Black RJ, Friedman RM, et al.: Systemic lupus erythematosus: presence in human serum of an unusual acid-labile leukocyte interferon. Science 1982, 216:429–431.
Rich SA: Human lupus inclusions and interferon. Science 1981, 213:772–775.
Ronnblom LE, Alm GV, Oberg KE: Possible induction of systemic lupus erythematosus by interferon-alpha treatment in a patient with a malignant carcinoid tumour. J Intern Med 1990, 227:207–210.
Gota C, Calabrese L: Induction of clinical autoimmune disease by therapeutic interferon-alpha. Autoimmunity 2003, 36:511–518.
Sigurdsson S, Nordmark G, Goring HH, et al.: Polymorphisms in the tyrosine kinase 2 and interferon regulatory factor 5 genes are associated with systemic lupus erythematosus. Am J Hum Genet 2005, 76:528–537. Genetic support for an association between polymorphisms in IRF5 and Tyk2 and SLE.
Blanco P, Palucha AK, Gill M, et al.: Induction of dendritic cell differentiation by IFN-alpha in systemic lupus erythematosus. Science 2001, 294:1540–1543.
Le BonA, Schiavoni G, D’Agostino G, et al.: Type I interferons potently enhance humoral immunity and can promote isotype switching by stimulating dendritic cells in vivo. Immunity 2001, 14:461–470.
Jego G, Palucka AK, Blanck JP, et al.: Plasmacytoid dendritic cells induce plasma cell differentiation through type I interferon and interleukin 6. Immunity 2003, 19:225–234.
Baechler EC, Batliwalla FM, Karypis G, et al.: Interferon-inducible gene expression signature in peripheral blood cells of patients with severe lupus. Proc Natl Acad Sci U S A 2003, 100:2610–2615. A large study of microarray gene expression analysis of PBMC from adult SLE patients demonstrating an IFN signature.
Baccala R, Kono DH, Theofilopoulos AN: Interferons as patho-genic effectors in autoimmunity. Immunol Rev 2005, 204:9–26.
Santiago-Raber ML, Baccala R, Haraldsson KM, et al.: Type-I interferon receptor deficiency reduces lupus-like disease in NZB Mice. J Exp Med 2003, 197:777–788. In vivo support for a contribution of the type I IFN pathway to autoimmunity and disease in murine lupus.
Braun D, Geraldes P, Demengeot J: Type I interferon controls the onset and severity of autoimmune manifestations in lpr mice. J Autoimmun 2003, 20:15–25.
Mathian A, Weinberg A, Gallegos M, et al.: IFN-alpha induces early lethal lupus in preautoimmune (New Zealand Black x New Zealand White) F1 but not in BALB/c mice. J Immunol 2005, 174:2499–2506.
Brinkmann V, Geiger T, Alkan S, Heusser CH: Interferon alpha increases the frequency of interferon gamma-producing human CD4+ T cells. J Exp Med 1993, 178:1655–1663.
Peng SL, Moslehi J, Craft J: Roles of interferon-gamma and inter-leukin-4 in murine lupus. J Clin Invest 1997, 99:1936–1946.
Haas C, Ryffel B, Le Hir M: IFN-gamma is essential for the development of autoimmune glomerulonephritis in MRL/lpr mice. J Immunol 1997, 158:5484–5491.
Balomenos D, Rumold R, Theofilopoulos AN: Interferon-gamma is required for lupus-like disease and lymphoaccu-mulation in MRL-lpr mice. J Clin Invest 1998, 101:364–371.
Schwarting A, Wada T, Kinoshita K, et al.: IFN-gamma receptor signaling is essential for the initiation, acceleration, and destruction of autoimmune kidney disease in MRL-Fas(lpr) mice. J Immunol 1998, 161:494–503.
Nicoletti F, Di Marco R, Zaccone P, et al.: Dichotomic effects of IFN-g on the development of systemic lupus erythematosus-like syndrome in MRL-lpr/lpr mice. Eur J Immunol 2000, 30:438.
Ozmen L, Roman D, Fountoulakis M, et al.: Experimental therapy of systemic lupus erythematosus: the treatment of NZB/W mice with mouse soluble interferon-gamma receptor inhibits the onset of glomerulonephritis. Eur J Immunol 1995, 25:6–12.
Lawson BR, Prud’homme GJ, Chang Y, et al.: Treatment of murine lupus with cDNA encoding IFN-gammaR/Fc. J Clin Invest 2000, 106:207–215.
Kirou KA, Lee C, George S, et al.: Coordinate overexpression of interferon-alpha-induced genes in systemic lupus erythematosus. Arthritis Rheum 2004, 50:3958–3967.
Kirou KA, Lee C, George S, et al.: Interferon-alpha pathway activation identifies a subgroup of systemic lupus erythema-tosus patients with distinct serologic features and active disease. Arthritis Rheum 2005, 52:1491–1503. Quantitative analysis of IFN-inducible gene expression in SLE PBMC demonstrating that IFN pathway activation defines a subgroup of patients with active disease. In addition, a strong association is noted between IFN pathway activation and the presence of autoantibodies specific for RNA-binding proteins.
Bennett L, Palucka AK, Arce E, et al.: Interferon and granu-lopoiesis signatures in systemic lupus erythematosus blood. J Exp Med 2003, 197:711–723.
Han GM, Chen SL, Shen N, et al.: Analysis of gene expression profiles in human systemic lupus erythematosus using oligonucleotide microarray. Genes Immun 2003, 4:177–186.
Crow MK, Wohlgemuth J: Microarray analysis of interferon-regulated genes in SLE. Arthritis Res Ther 2003, 5:279–287.
Crow MK, Kirou KA, Wohlgemuth J: Microarray of interferon-regulated genes in SLE. Autoimmunity 2003, 36:481–490.
Khabar KS, Al-Haj L, Al-Zoghaibi F, et al.: Expressed gene clusters associated with cellular sensitivity and resistance towards anti-viral and anti-proliferative actions of interferon. J Mol Biol 2004, 342:833–846.
Magnusson M, Magnusson S, Vallin H, et al.: Importance of CpG dinucleotides in activation of natural IFN-alpha-producing cells by a lupus-related oligodeoxynucleotide. Scand J Immunol 2001, 54:543–550.
Vallin H, Peters A, Alm GV, Ronnblom L: Anti-double-stranded DNA antibodies and immunostimulatory plasmid DNA in combination mimic the endogenous IFN-alpha inducer in systemic lupus erythematosus. J Immunol 1999, 163:6306–6313.
Bave U, Alm GV, Ronnblom L: The combination of apoptotic U937 cells and lupus IgG is a potent IFN-alpha inducer. J Immunol 2000, 165:3519–3526.
Bave U, Vallin H, Alm GV, Ronnblom L: Activation of natural interferon-alpha producing cells by apoptotic U937 cells combined with lupus IgG and its regulation by cytokines. J Autoimmun 2001, 17:71–80.
Lovgren T, Eloranta ML, Bave U, et al.: Induction of interferon-alpha production in plasmacytoid dendritic cells by immune complexes containing nucleic acid released by necrotic or late apoptotic cells and lupus IgG. Arthritis Rheum 2004, 50:1861–1772. In vitro data demonstrating the capacity of RNA and DNA components in SLE serum, along with antibodies, to induce IFN-á production in PDCs.
Svensson H, Johannisson A, Nikkila T, et al.: The cell surface phenotype of human natural interferon-a producing cells as determined by flow cytometry. Scand J Immunol 1996, 44:164–172.
Siegel FP, Kadowaki N, Shodell M, et al.: The nature of the principal type I interferon-producing cells in human blood. Science 1999, 284:1835–1837.
Blomberg S, Eloranta ML, Magnusson M, et al.: Expression of the markers BDCA-2 and BDCA-4 and production of interferon-alpha by plasmacytoid dendritic cells in systemic lupus erythematosus. Arthritis Rheum 2003, 48:2524–2532.
Leadbetter EA, Rifkin IR, Hohlbaum AM, et al.: Chromatin-IgG complexes activate B cells by dual engagement of IgM and Toll-like receptors. Nature 2002, 416:603–637. Demonstration in a murine in vitro system of B cell activation induced by immune complexes containing CpG DNA oligonucleotides.
Viglianti GA, Lau CM, Hanley TM, et al.: Activation of auto-reactive B cells by CpG dsDNA. Immunity 2003, 19:837–847.
Christensen SR, Kashgarian M, Alexopoulou L, et al.: Toll-like receptor 9 controls anti-DNA autoantibody production in murine lupus. J Exp Med 2005, 202:321–331. Demonstration, in a murine system, that Toll-like receptor 9 is required for anti-DNA antibody production. Importantly, production of anti-Sm antibody was intact or increased in mice deficient in TLR9, and absence of TLR9 and anti-DNA autoantibodies did not abrogate nephritis. The data presented in this paper suggest a complex set of pathogenic mechanisms in which distinct TLRs may contribute to autoimmunity and disease, with no one receptor in itself responsible.
Means TK, Latz E, Hayashi F, et al.: Human lupus autoanti-body-DNA complexes activate DCs through cooperation of CD32 and TLR9. J Clin Invest 2005, 115:407–417.
Scheinecker C, Zwolfer B, Koller M, et al.: Alterations of den-dritic cells in systemic lupus erythematosus: phenotypic and functional deficiencies. Arthritis Rheum 2001, 44:856–865.
Blomberg S, Eloranta ML, Cederblad B, et al.: Presence of cutaneous interferon-alpha producing cells in patients with systemic lupus erythematosus. Lupus 2001, 10:484–490.
Farkas L, Beiske K, Lund-Johansen F, et al.: Plasmacytoid dendritic cells (natural interferon-alpha/beta -producing cells) accumulate in cutaneous lupus erythematosus lesions. Am J Pathol 2001, 159:237–243.
Chuntharapai A, Lai J, Huang X, et al.: Characterization and humanization of a monoclonal antibody that neutralizes human leukocyte interferon: a candidate therapeutic for IDDM and SLE. Cytokine 2001, 15:250–260.
Duramad O, Fearon KL, Chang B, et al.: Inhibitors of TLR-9 act on multiple cell subsets in mouse and man in vitro and prevent death in vivo from systemic inflammation. J Immunol 2005, 174:5193–5200.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Crow, M.K. Interferon pathway activation in systemic lupus erythematosus. Curr Rheumatol Rep 7, 463–468 (2005). https://doi.org/10.1007/s11926-005-0053-4
Issue Date:
DOI: https://doi.org/10.1007/s11926-005-0053-4