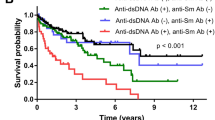
Abstract
Autoimmune rheumatic diseases (ARD), such as systemic lupus erythematosus (SLE), rheumatoid arthritis (RA) and Sjögren's syndrome (SS), have consistently been associated with the development of B-cell non-Hodgkin lymphoma (BCNHL). In this Review, we focus on reports published since 2006 and summarize the data regarding the BCNHL subtypes and clinical findings associated with this increased risk. Patients with these ARD, particularly those with detectable autoantibodies and systemic involvement, are at increased risk of developing BCNHL, especially diffuse large B-cell lymphoma and marginal zone lymphoma. SS shows the strongest association with BCNHL. Male sex, advanced age, prolonged disease course and increased disease severity, but not family history of autoimmune conditions, seem to be associated with an increased risk of non-Hodgkin lymphoma. Chronic immune stimulation, genetic and environmental factors and some immunosuppressive drugs might be involved in lymphomagenesis in these patients. The reason why some ARD are associated with BCNHL and other autoimmune diseases are not remains unclear. These associations are important as they provide information about the mechanisms of lymphomagenesis, and might help identify new therapeutic targets.
Key Points
-
Patients with systemic lupus erythematosus (SLE), rheumatoid arthritis (RA) and Sjögren's syndrome (SS) are at increased risk of developing B-cell non-Hodgkin lymphoma, with the highest risk associated with SS
-
The subtypes most strongly associated with SS and SLE are diffuse large B-cell lymphoma (DLBCL) and marginal zone lymphoma, whereas DLBCL and lymphoplasmacytic lymphoma are most common in RA
-
Chronic autoimmune and pathogen-induced immune stimulation, genetic and environmental factors and some immunosuppressant therapies are likely to be involved in lymphomagenesis
-
Some drugs that target B cells have been shown to be effective against both autoimmune rheumatic diseases and lymphomas
-
The clinical benefits obtained with these drugs demonstrate the importance of B cells in the pathogenesis of these diseases, and has led to the development of new drugs of this class
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Jaffe, E. S., Harris, N. L., Stein, H. & Isaacson, P. G. Classification of lymphoid neoplasms: the microscope as a tool for disease discovery. Blood 112, 4384–4399 (2008).
Grulich, A. E. & Vajdic, C. M. The epidemiology of non-Hodgkin lymphoma. Pathology 37, 409–419 (2005).
Goldin, L. R. & Landgren, O. Autoimmunity and lymphomagenesis. Int. J. Cancer 124, 1497–1502 (2009).
Bierman, P. J., Harris, N. & Armitage, J. O. in Cecil Medicine 23rd edn 1408–1420 (Saunders Elsevier, Philadelphia, 2007).
Longo, D. L. in Harrison's Principles of Internal Medicine 17th edn 687–688 (McGraw-Hill Medical, New York, 2008).
Zintzaras, E., Voulgarelis, M. & Moutsopoulos, H. M. The risk of lymphoma development in autoimmune diseases: a meta-analysis. Arch. Intern. Med. 165, 2337–2344 (2005).
Alexander, D. D. et al. The non-Hodgkin lymphomas: a review of the epidemiologic literature. Int. J. Cancer 120 (Suppl.), 1–39 (2007).
Kiss, E., Kovacs, L. & Szodoray, P. Malignancies in systemic lupus erythematosus. Autoimmun. Rev. 9, 195–199 (2010).
Hansen, A., Lipsky, P. E. & Dörner, T. B-cell lymphoproliferation in chronic inflammatory rheumatic diseases. Nat. Clin. Pract. Rheumatol. 3, 561–569 (2007).
Baecklund, E. et al. Association of chronic inflammation, not its treatment, with increased lymphoma risk in rheumatoid arthritis. Arthritis Rheum. 54, 692–701 (2006).
Smedby, K. E. et al. Autoimmune and chronic inflammatory disorders and risk of non-Hodgkin lymphoma by subtype. J. Natl Cancer Inst. 98, 51–60 (2006).
Smedby, K. E. et al. Autoimmune disorders and risk of non-Hodgkin lymphoma subtypes: a pooled analysis within the InterLymph Consortium. Blood 111, 4029–4038 (2008).
Mellemkjaer, L. et al. Autoimmune disease in individuals and close family members and susceptibility to non-Hodgkin's lymphoma. Arthritis Rheum. 58, 657–666 (2008).
Anderson, L. A. et al. Population-based study of autoimmune conditions and the risk of specific lymphoid malignancies. Int. J. Cancer 125, 398–405 (2009).
Ansell, P. et al. Non-Hodgkin lymphoma and autoimmunity: does gender matter? Int J Cancer. doi: 10.1002/ijc.25680.
Jemal, A. et al. Cancer statistics, 2009. CA Cancer J. Clin. 59, 225–249 (2009).
Smith, A. et al. The Haematological Malignancy Research Network (HMRN): a new information strategy for population based epidemiology and health service research. Br. J. Haematol. 148, 739–753 (2010).
Kristinsson, S. Y. et al. Immune-related and inflammatory conditions and risk of lymphoplasmacytic lymphoma or Waldenström macroglobulinemia. J. Natl Cancer Inst. 102, 557–567 (2010).
Bernatsky, S. et al. An international cohort study of cancer in systemic lupus erythematosus. Arthritis Rheum. 52, 1481–1490 (2005).
Tincani, A., Taraborelli, M. & Cattaneo, R. Antiphospholipid antibodies and malignancies. Autoimmun. Rev. 9, 200–202 (2010).
Hellgren, K., Smedby, K. E., Feltelius, N., Baecklund, E. & Askling, J. Do rheumatoid arthritis and lymphoma share risk factors? A comparison of lymphoma and cancer risks before and after diagnosis of rheumatoid arthritis. Arthritis Rheum. 62, 1252–1258 (2010).
Lazarus, M. N., Robinson, D., Mak, V., Moller, H. & Isenberg, D. A. Incidence of cancer in a cohort of patients with primary Sjögren's syndrome. Rheumatology 45, 1012–1015 (2006).
Geborek, P. et al. Tumor necrosis factor do not increase overall risk in patient with rheumatoid arthritis, but may be associated with an increased risk of lymphomas. Ann. Rheum. Dis. 64, 657–658 (2005).
De Vita, S. & Quartuccio, L. Treatment of rheumatoid arthritis with rituximab: an update and possible indications. Autoimmun. Rev. 5, 443–448 (2006).
Mariette, X. et al. Lymphoma in patients treated with anti-TNF: results of the 3-year prospective French RATIO registry. Ann. Rheum. Dis. 69, 400–408 (2010).
Askling, J. et al. Cancer risk with tumor necrosis factor alpha (TNF) inhibitors: meta-analysis of randomized controlled trials of adalimumab, etanercept, and infliximab using patient level data. Pharmacoepidemiol. Drug Saf. 20, 119–130 (2011).
Kovács, L., Szodoray, P. & Kiss, E. Secondary tumours in Sjögren's Syndrome. Autoimmun. Rev. 9, 203–206 (2010).
Bende, R. J., van Maldegem, F. & van Noesel, C. J. Chronic inflammatory disease, lymphoid tissue neogenesis and extranodal marginal zone B-cell lymphomas. Haematologica 94, 1109–1123 (2009).
Kristinsson, S. Y. et al. Genetic and immune-related factors in the pathogenesis of lymphoproliferative and plasma cell malignancies. Haematologica 94, 1581–1589 (2009).
Goodnow, C. C. Multistep pathogenesis of autoimmune disease. Cell 130, 25–35 (2007).
Youinou, P. Is BAFF the murderer in lupus? Lupus 17, 613–614 (2008).
Tangye, S. G., Bryant, V. L., Cuss, A. K. & Good, K. L. BAFF, APRIL and human B cell disorders. Semin. Immunol. 18, 305–317 (2006).
Cellier, C. et al. Refractory sprue, coeliac disease, and enteropathy-associated T-cell lymphoma. French Coeliac Disease Study Group. Lancet 356, 203–208 (2000).
Suarez, F., Lortholary, O., Hermine, O. & Lecuit, M. Infection-associated lymphomas derived from marginal zone B cells: a model of antigen-driven lymphoproliferation. Blood 107, 3034–3044 (2006).
Bernatsky, S., Ramsey-Goldman, R. & Clarke, A. E. Malignancy in systemic lupus erythematosus: what have we learned? Best Pract. Res. Clin. Rheumatol. 23, 539–547 (2009).
Ramos-Casals, M. et al. Characterization of B cell lymphoma in patients with Sjogren's syndrome and hepatitis C virus infection. Arthritis Rheum 57, 161–170 (2007).
Brady, G., Macarthur, G. J. & Farrell, P. J. Epstein–Barr virus and Burkitt lymphoma. Postgrad. Med. J. 84, 372–377 (2008).
Calza, L. et al. Systemic and discoid lupus erythematosus in HIV-infected patients treated with highly active antiretroviral therapy. Int. J. STD AIDS 14, 356–359 (2003).
Reyes, S. L. I. et al. BAFF: a regulatory cytokine of B lymphocytes involved in autoimmunity and lymphoid cancer [Spanish]. Rev. Med. Chil. 134, 1175–1184 (2006).
Wang, S. S. et al. Immune mechanisms in non-Hodgkin lymphoma: joint effects of the TNF G308A and IL10 T3575A polymorphisms with non-Hodgkin lymphoma risk factors. Cancer Res. 67, 5042–5054 (2007).
Bayley, J. P., Ottenhoff, T. H. & Verweij, C. L. Is there a future for TNF promoter polymorphisms? Genes Immun. 5, 315–329 (2004).
Gibson, A. W. et al. Novel single nucleotide polymorphisms in the distal IL-10 promoter affect IL-10 production and enhance the risk of systemic lupus erythematosus. J. Immunol. 166, 3915–3922 (2001).
Balkwill, F. & Coussens, L. M. Cancer: an inflammatory link. Nature 431, 405–406 (2004).
Wang, S. S. et al. Common genetic variants in proinflammatory and other immunoregulatory genes and risk for non-Hodgkin lymphoma. Cancer Res. 66, 9771–9780 (2006).
Chen, Y. et al. Cytokine polymorphisms in Th1/Th2 pathway genes, body mass index and risk of non-Hodgkin lymphoma. Blood 117, 585–589 (2011).
Toussirot, E. & Wendling, D. The use of TNF-α blocking agents in rheumatoid arthritis: an update. Expert Opin. Pharmacother. 8, 2089–2107 (2007).
Bernatsky, S. et al. The relationship between cancer and medication exposures in systemic lupus erythematosus: a case–cohort study. Ann. Rheum. Dis. 67, 74–79 (2008).
Dörner, T. & Lipsky, P. E. B-cell targeting: a novel approach to immune intervention today and tomorrow. Expert Opin. Biol. Ther. 7, 1287–1299 (2007).
Dörner, T. et al. Current status on B-cell depletion therapy in autoimmune diseases other than rheumatoid arthritis. Autoimmun. Rev. 9, 82–89 (2009).
Dörner, T., Kinnman, N. & Tak, P. P. Targeting B cells in immune-mediated inflammatory disease: a comprehensive review of mechanisms of action and identification of biomarkers. Pharmacol. Ther. 125, 464–475 (2010).
Quartuccio, L. et al. Controversies on rituximab therapy in Sjögren syndrome-associated lymphoproliferation. Int. J. Rheumatol. 2009, 424935 (2009).
Edwards, J. C., Leandro, M. J. & Cambridge, G. Repeated B lymphocyte depletion therapy in rheumatoid arthritis: 5 year follow-up. Arthritis Rheum. 52 (Suppl.), S133–S134 (2005).
Popa, C., Leandro, M. J., Cambridge, G. & Edwards, J. C. Repeated B lymphocyte depletion with rituximab in rheumatoid arthritis over 7 yrs. Rheumatology (Oxford) 46, 626–630 (2007).
Watanabe, T. Treatment strategies for nodal and gastrointestinal follicular lymphoma: current status and future development. World J. Gastroenterol. 16, 5543–5554 (2010).
Voulgarelis, M., Giannouli, S., Tzioufas, A. G. & Moutsopoulos, H. M. Long term remission of Sjögren's syndrome associated aggressive B cell non-Hodgkin's lymphomas following combined B cell depletion therapy and CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone). Ann. Rheum. Dis. 65, 1033–1037 (2006).
Voulgarelis, M., Giannouli, S., Anagnostou, D. & Tzioufas, A. G. Combined therapy with rituximab plus cyclophosphamide/doxorubicin/vincristine/prednisone (CHOP) for Sjögren's syndrome-associated B-cell aggressive non-Hodgkin's lymphomas. Rheumatology (Oxford) 43, 1050–1053 (2004).
Pijpe, J. et al. Rituximab treatment in patients with primary Sjögren's syndrome: an open-label phase II study. Arthritis Rheum. 52, 2740–2750 (2005).
Favas, C. & Isenberg, D. A. B-cell depletion therapy in SLE—what are the current prospects for its acceptance? Nat. Rev. Rheumatol. 5, 711–716 (2009).
Morschhauser, F. et al. Humanized anti-CD20 antibody, veltuzumab, in refractory/recurrent non-Hodgkin's lymphoma: phase I/II results. J. Clin. Oncol. 27, 3346–3353 (2009).
Milani, C. & Castillo, J. Veltuzumab, an anti-CD20 mAb for the treatment of non-Hodgkin's lymphoma, chronic lymphocytic leukemia and immune thrombocytopenic purpura. Curr. Opin. Mol. Ther. 11, 200–207 (2009).
Steinfeld, S. D. et al. Epratuzumab (humanized anti-CD22 antibody) in primary Sjögren's syndrome: an open-label phase I/II study. Arthritis Res. Ther. 8, R129 (2006).
Author information
Authors and Affiliations
Contributions
C. Dias researched data for and wrote the article. D. A. Isenberg discussed the content of the article and performed review/editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Dias, C., Isenberg, D. Susceptibility of patients with rheumatic diseases to B-cell non-Hodgkin lymphoma. Nat Rev Rheumatol 7, 360–368 (2011). https://doi.org/10.1038/nrrheum.2011.62
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2011.62
This article is cited by
-
Long-term follow-up in primary Sjögren’s syndrome reveals differences in clinical presentation between female and male patients
Biology of Sex Differences (2017)
-
Acceleration of tumor growth due to dysfunction in M1 macrophages and enhanced angiogenesis in an animal model of autoimmune disease
Laboratory Investigation (2016)
-
Targeting Bruton's tyrosine kinase in B cell malignancies
Nature Reviews Cancer (2014)
-
An unusual case of glomerulonephritis in a patient with non-Hodgkin Mucosal Associated Lymphoid Tissue (MALT) B-cell lymphoma
BMC Nephrology (2013)
-
Malignancies in children and young adults on etanercept: summary of cases from clinical trials and post marketing reports
Pediatric Rheumatology (2013)